GEXVal Completes FRAXA-DVI, Receives FDA Orphan Drug Designation, and Prepares for Phase 2a Trial
Orphan Drug Designation positions GXV-001 for next steps after FRAXA-DVI completion as GEXVal prepares a Phase 2a Fragile X trial in Europe.
Nospharma Announces Partnership with FRAXA to Test NOS-01 in Pre-Clinical Models of Fragile X
NOS-01 enters the FRAXA Drug Validation Initiative (FRAXA-DVI) for Fragile X mouse-model testing, generating data to guide development decisions.
FRAXA Drug Validation Initiative (FRAXA-DVI)
The FRAXA Drug Validation Initiative (FRAXA-DVI) provides streamlined, cost-effective preclinical testing to evaluate investigational and repurposed compounds for Fragile X syndrome.
BK Channel Openers: From FRAXA Seed Funding to Big Pharma Investment and Trials
See how FRAXA’s early grants propelled Fragile X research from lab discoveries to industry momentum, with partners advancing therapies into trials.
GEXVal and FRAXA Collaborate to Advance Fragile X Research with Phase 2a Trial
GEXVal and FRAXA collaborate to advance Fragile X research with the Phase 2a trial of GXV-001, supported by AMED’s funding program.
BK Channel Openers: A New Hope for Fragile X Treatment – Insights from Kaerus Bioscience CEO Robert Ring
Kaerus Bioscience’s BK channel openers for Fragile X syndrome are advancing through Phase 1 trials, offering hope for new treatments with FRAXA’s continued support.
Marvel Biosciences Partners with FRAXA to Test MB204 for Fragile X Syndrome
Marvel Biosciences and FRAXA Research Foundation are collaborating to test MB204, a promising new treatment for Fragile X which targets adenosine receptors.
Fragile X Treatment Target Emerges from Neurolixis & FRAXA Collaboration
FRAXA pharma partner Neurolixis has filed patents for treatment of Fragile X with a highly selective new drug, NLX-101, which targets the serotonin receptor 5-HT1A.
BK Channel Openers: A New Drug for Fragile X Is Ready for Clinical Trials
A promising new BK channel opener, SPG601 from Spinogenix, is entering clinical trials for Fragile X syndrome. Learn about its potential to restore synaptic function and address core symptoms.
Inside the FRAXA Drug Validation Initiative: Advancing Fragile X Treatments
FRAXA-DVI is revolutionizing Fragile X syndrome research, providing efficient, comprehensive and objective preclinical testing of potential treatments.
Two-Med Combo Normalized Behavior, Improved Memory in Fragile X Mice
Treating Fragile X might require a combination of drugs. FRAXA-DVI tested ibudilast and gaboxadol in Fragile X mice. Together they rescued a wide array of symptoms.
Coming Together for Rare Disease Day 2023
Today we mark Rare Disease Day. FRAXA is committed to advancing research on Fragile X, one of the most common rare diseases worldwide.
Fragile X Clinical Trial of New PDE4D Inhibitor from Tetra
A $200K FRAXA grant enabled a successful Phase 2 trial of a PDE4D inhibitor for adult men with Fragile X, showing strong cognitive gains without side effects or tolerance.
New Fragile X Clinical Trial Announced by Healx
Healx has received Investigational New Drug (IND) approval from the US Food and Drug Administration (FDA) for a Phase 2a clinical study of HLX-0201 (sulindac, an FDA-approved drug).
Purposeful and FRAXA Partnership Leads to Clinical Trial
AI and FRAXA-DVI identified a drug + supplement combo that reversed all Fragile X symptoms in mice. A clinical trial tested this in adults with Fragile X.
Making Drug Development Efficient Through Community-Based Collaboration
FRAXA’s partnership with Anavex shows how early collaboration between patient advocates and pharma can accelerate drug development for Fragile X and rare diseases.
Tetra’s Fragile X Clinical Trial – The Most Successful So Far
Dr. Mark Gurney of Tetra Therapeutics, discusses how one of the earliest clues to the biology of Fragile X led to the most successful Fragile X clinical trial to date: a phase 2 trial of a PDE4D inhibitor.
Promising Results of Preclinical Study of ANAVEX®2-73
Anavex Life Sciences announced today that preclinical data of the ANAVEX®2-73 (blarcamesine) study in Fragile X syndrome were published in the journal, Scientific Reports.
Synaptogenix Announced Intention to Launch a Fragile X Clinical Trial with Bryostatin
Bryostatin research has advanced from mouse models to human trials. Synaptogenix and Nemours make plans to test this potential treatment in Fragile X clinical trials.
Tetra Releases Full Results of FRAXA-Funded Clinical Trial of PDE4D Inhibitor
Today, Tetra Therapeutics published the full results of its PDE4D trial. The data from this phase 2 clinical trial suggests the PDE4D drug can improve intellectual disability in Fragile X syndrome.
Bryostatin-1 in Long-term Use Seen to Arrest Fragile X Symptoms in Mouse Model
Long-term, but not short-term, treatment with bryostatin-1 arrested some behavioral and cognitive symptoms in a mouse model of Fragile X.
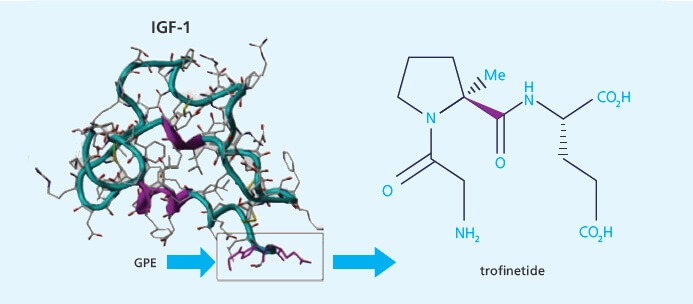
Trofinetide Clinical Trial Results Published
Neuren Pharmaceuticals has published results of a Phase 2 Fragile X Clinical Trial of Trofinetide ih Fragile X syndrome. This trial tested a new design which can be used in future trials.
Healx Raises $56M to use AI to Find Treatments for Fragile X & Other Rare Diseases
Healx has secured $56M in new financing to build a clinical-stage portfolio for rare diseases, including treatments for Fragile X syndrome, and to launch a global Rare Treatment Accelerator program. Where the traditional drug discovery model takes more than a decade and can run into the billions of dollars, Healx’s AI-driven approach makes the process faster, more efficient and cost-effective.
Tetra Announces $40M to Advance BPN14770 for FXS and Alzheimer’s Disease
Tetra conducted a Phase 2 study of BPN14770 in adults with Fragile X Syndrome. This clinical trial was made possible by early work with the FRAXA-DVI and over $200,000 from FRAXA.