10 Year Vision for Fragile X Research – Dr. Elizabeth Berry-Kravis & Dr. Patricia Cogram
FRAXA Investigators Dr. Patricia Cogram and Dr. Elizabeth Berry-Kravis reflect on progress that has been made and visualize what they see happening in the next 10 years for people living with Fragile X syndrome.
Clinical Trial Results Reported for Phase 3 CONNECT-FX Study of Zygel™
Disappointing results have been published from Zynerba Pharmaceuticals’s phase 3 clinical trial of Zygel™. 212 children and adolescents 3 to 17 years were given Zygel or placebo for 12 weeks.
FRAXA Volunteer Participates in Peer Reviewed Medical Research Program for the Department of Defense
FRAXA advocate Jennifer Frobish served as a reviewer for the Department of Defense’s medical research program, evaluating Fragile X–related proposals.
Why Pharma Companies Take on Fragile X, Explained
Research aimed at finding Fragile X syndrome treatments is exploding. Why are so many companies investing? FRAXA CSO Dr. Michael Tranfaglia explains many reasons Fragile X is a hot topic.
FMR1 Renamed to Fragile X Messenger Ribonucleoprotein 1
The FMR1 gene is renamed to “Fragile X Messenger Ribonucleoprotein 1” gene. The Fragile X protein, FMRP, is "Fragile X Messenger Ribonucleoprotein." "Mental retardation" is not used.
How FRAXA Prioritizes Research, Explained
Dr. Mike Tranfaglia explains how FRAXA prioritizes research. "It’s not either-or. It’s not we have a definitive treatment or we have a new drug treatment or we have a repurposing treatment.
Aggression, Other Fragile X Behaviors Tend to Ease Over Teenage Years
Behavioral problems such as hyperactivity and aggression are more frequent in children with Fragile X syndrome, becoming less common as they grow through adolescence into adulthood.
What FRAXA Is Excited about in the Upcoming Fragile X Research Grants, Explained
Many new clinical trials are starting, with existing and investigational drugs and other treatment strategies that have been tested in the Fragile X mouse models moving to patients in clinical trials.
Gordon Research Conference for Fragile X and Autism-Related Disorders, Explained
The Fragile X and Autism-Related Disorders Gordon Research Conference is a biannual event, an international forum for the presentation and discussion of frontier research in these conditions.
Potential Upcoming Advances in Fragile X Research
Dr. Peter Kind and Dr. Nahum Sonenberg share their optimism about the next 10 years of Fragile X research. They discuss where they think the next big discoveries will emerge.
Neurodevelopmental Drug Development Summit Presentation
FRAXA president and co-founder, Katie Clapp, and and FRAXA Scientific Advisor, Dr. Elizabeth Berry-Kravis, presented at the Neurodevelopmental Drug Development Summit.
Reactivating the Fragile X Gene in Young Mice Reverses Symptoms
Fragile X syndrome might be treated by reactivating the gene which is shut down in the syndrome. Researchers were able to reduce FXS symptoms by inserting the FMR1 gene into the brains of young mice.
10 Year Vision for Collaborations That Transform Fragile X and Autism Research
FRAXA’s Dr. Mike Tranfaglia talks with Autism Science Foundation’s Allison Singer about collaborations between the Fragile X community and other disability organizations as we look to the next 10 years.
New Fragile X Clinical Trial Announced by Healx
Healx has received Investigational New Drug (IND) approval from the US Food and Drug Administration (FDA) for a Phase 2a clinical study of HLX-0201 (sulindac, an FDA-approved drug).
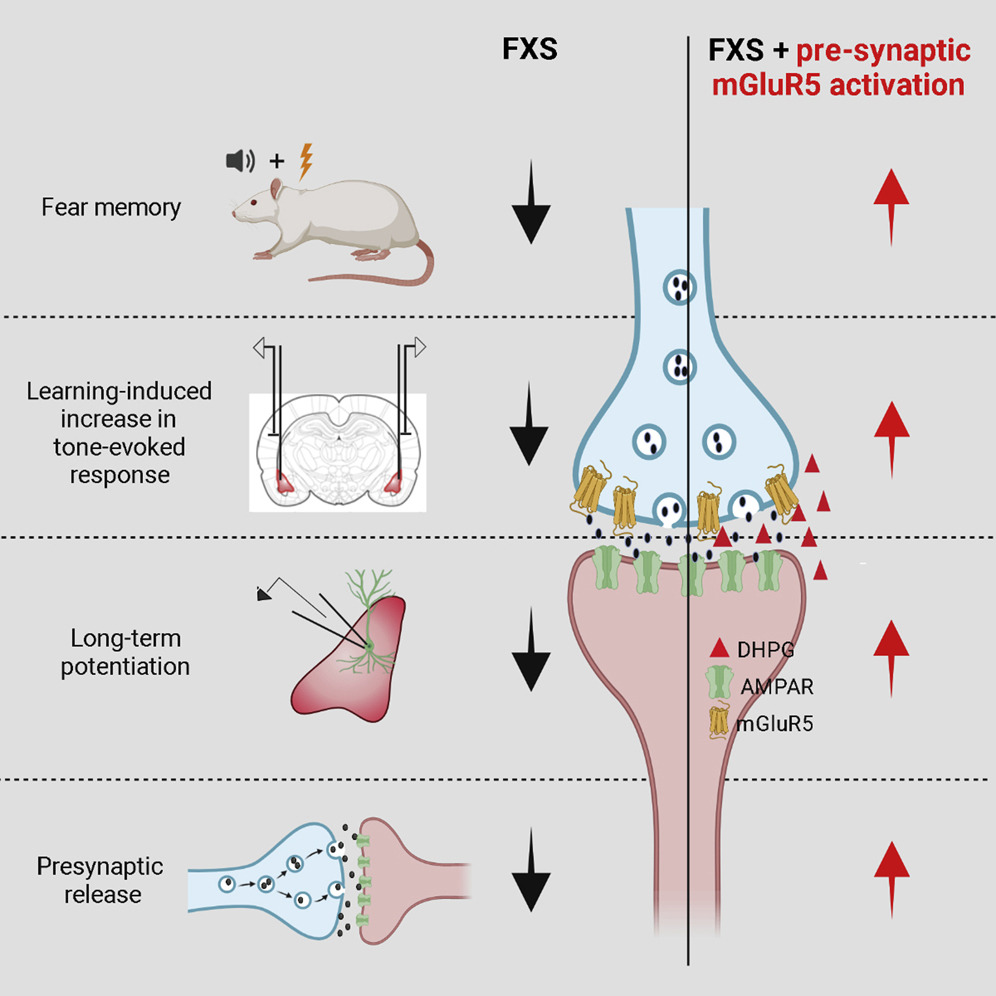
Correcting the Brain’s Emotional Memory Center
FRAXA Investigator Dr. Sumatra Chattarji investigated the synaptic basis of deficient conditioned fear and its reversal in Fragile X syndrome rats. Results published.
Making Drug Development Efficient Through Community-Based Collaboration
FRAXA’s partnership with Anavex shows how early collaboration between patient advocates and pharma can accelerate drug development for Fragile X and rare diseases.
2021 Fragile X Research Grants Funded by FRAXA Research Foundation
Each year, FRAXA funds a diverse portfolio of research. FRAXA Fellowships and Grants are seed funding for the future, the feedstock for the Fragile X treatment development pathway.
20 Years of Advancing Fragile X Research: Progress Toward a Cure
MIT Professor Mark Bear traces the discoveries that give us great optimism of finding effective treatments and ultimately a cure for Fragile X syndrome.
Brain Organoids, Moving Fragile X Research Forward
FRAXA-funded scientists at Emory created human brain organoids that reveal Fragile X changes more clearly than mouse models, opening new paths to targeted treatments.
Tetra’s Fragile X Clinical Trial – The Most Successful So Far
Dr. Mark Gurney of Tetra Therapeutics, discusses how one of the earliest clues to the biology of Fragile X led to the most successful Fragile X clinical trial to date: a phase 2 trial of a PDE4D inhibitor.
Promising Results of Preclinical Study of ANAVEX®2-73
Anavex Life Sciences announced today that preclinical data of the ANAVEX®2-73 (blarcamesine) study in Fragile X syndrome were published in the journal, Scientific Reports.
Synaptogenix Announced Intention to Launch a Fragile X Clinical Trial with Bryostatin
Bryostatin research has advanced from mouse models to human trials. Synaptogenix and Nemours make plans to test this potential treatment in Fragile X clinical trials.
Fragile X Syndrome: In Pursuit of a Cure Webinar
Global webinar “Fragile X Syndrome: In Pursuit of a Cure” on July 22, 2021 commemorated World Fragile X Day. Over 5,000 registered from more than 50 countries.
Drug Tolerance in MGluR5 Clinical Trials – Dr Patrick McCamphill 1:1 with FRAXA
We suspected that clinical trials of mGluR5 blockers from Novartis and Roche failed because the drug triggered tolerance. Dr. Patrick does find tolerance, and is looking for ways to overcome it.