Fragile X syndrome research shows PDE inhibitors may improve brain function and behavior, with promise for related neurodevelopmental disorders.
Read moreTetra Therapeutics
Shionogi’s EXPERIENCE Phase 3 Clinical Trial of Zatolmilast in Fragile X Syndrome
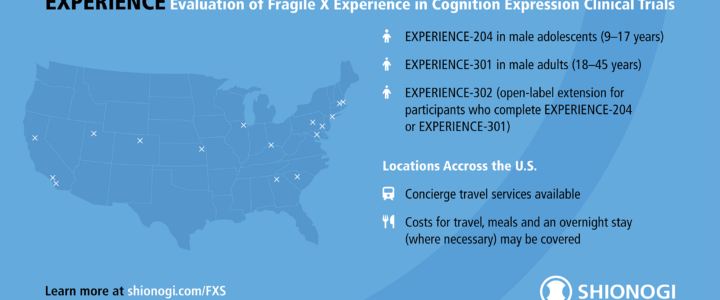
Shionogi’s EXPERIENCE clinical trials for Fragile X syndrome are nearing completion. Enrollment for the adult trial (EXPERIENCE-301) is now closed, while the adolescent trial (EXPERIENCE-204) is in its final phase. Learn more about the study, FRAXA’s role, and the open-label extension.
Read moreNPR Spotlights Zatolmilast: A Potential Breakthrough for Fragile X Syndrome

NPR spotlights zatolmilast, a promising drug offering new hope for individuals with Fragile X syndrome. Families report life-changing improvements in anxiety, communication, and independence.
Read moreExploring Advances Fragile X Research: Comprehensive Webinar Highlights – May 2024

Discover the latest in Fragile X research with insights from our May 2024 webinar. Learn about new FRAXA grants, key clinical trials, and scientific updates that are shaping the future of Fragile X syndrome treatments.
Read moreInside the FRAXA Drug Validation Initiative: Advancing Fragile X Treatments

Explore how the FRAXA Drug Validation Initiative is revolutionizing Fragile X syndrome treatment, leading the charge towards innovative therapies and hope for affected families.
Read moreTwo-Med Combo Normalized Behavior, Improved Memory in Fragile X Mice
Treating Fragile X syndrome will likely require a combination of drugs, as a single medication may not address all symptoms. At FRAXA-DVI, Dr. Patricia Cogram and her team recently tested a combination of two investigational new drugs in Fragile X mice, with support from Healx. Together ibudilast and gaboxadol rescued a wide array of symptoms in the mice.
Read moreASOs and Fragile X: Addressing the Most Asked Questions

Explore the potential of ASOs in treating Fragile X syndrome & FXTAS. Dive into a comprehensive Q&A addressing key questions and breakthrough findings.
Read moreComing Together for Rare Disease Day 2023

Today, February 28, we mark Rare Disease Day, a day dedicated to raising awareness about rare diseases and highlighting the need for continued research and collaboration. At FRAXA Research Foundation, we are committed to advancing research on Fragile X syndrome, one of the most common rare diseases worldwide.
Read moreFragile X Clinical Trial of New PDE4D Inhibitor from Tetra

With a $200,043 grant from FRAXA Research Foundation, Dr. Elizabeth Berry-Kravis completed a successful Phase 2 clinical trial of a PDE4 inhibitor for adult men with Fragile X syndrome. This trial treated 30 males, 18-45 years of age with a new PDE4D allosteric inhibitor from Tetra Discovery Partners using a crossover design, so that everyone got active drug for part of the time and placebo for part of the time.
Read moreTranscriptional Signatures Sensitive to Cognition-Improving Pharmacological Treatments in Fragile X Syndrome

The Fragile X field needs biomarkers to accurately measure the effects of potential treatments in both Fragile X mice and in humans. Dr. Ozaita and his team have found molecular features in the brain that can serve as an objective signature for the syndrome. They will use this tool to test cannabidiol and two other drugs in mice.
Read moreTetra’s Fragile X Clinical Trial – The Most Successful So Far

Dr. Mark Gurney, CEO of Tetra Therapeutics, discusses how one of the earliest clues to the biology of Fragile X led to the most successful Fragile X clinical trial to date. FRAXA and Tetra began working together after a key FRAXA-funded study caught the attention of Dr. Gurney. Through the FRAXA Drug Validation Initiative, Dr. Patricia Cogram was able to conduct preclinical validation experiments with Tetra’s lead compound in record time, paving the way for clinical trials.
Read moreFragile X Syndrome: In Pursuit of a Cure Webinar

A global webinar titled “Fragile X Syndrome: In Pursuit of a Cure,” took place on July 22, 2021 to commemorate World Fragile X Day. This complimentary event is co-organized with WuXi AppTec. We are delighted that more than 5,000 registered from more than 50 countries worldwide, coming together to raise awareness of Fragile X, and to foster collaborations towards effective treatments and ultimately a cure.
Read moreFRAXA Drug Validation Initiative (FRAXA-DVI)

The FRAXA Drug Validation Initiative (FRAXA-DVI) provides speedy, cost-effective, objective preclinical testing of potential Fragile X treatments. FRAXA-DVI uses in-vitro systems, behavior batteries, and gene expression and peripheral biomarker platforms to validate investigational new drugs and repurposed available compounds in Fragile X syndrome (FXS).
Read moreTetra Releases Full Results of FRAXA-Funded Clinical Trial of PDE4D Inhibitor

Today, Tetra Therapeutics published the full results of its PDE4D trial published the full results to their announcement. Now having reviewed the full results, FRAXA can confidently say that the PDE4D drug trial gives hope to patients and families that Fragile X Syndrome is a treatable disorder, and this particular drug can improve intellectual disability.
Read moreClinical Trials and Cyclic AMP in Fragile X Syndrome: A Life Journey

In November 2020, a phase II clinical trial reported extremely successful results. This clinical trial of a PDE4D inhibitor from Tetra Pharmaceuticals was conducted by Dr. Elizabeth Berry-Kravis at Rush University Medical Center and funded by FRAXA Research Foundation. In this Simons Foundation lecture, Elizabeth Berry-Kravis traces 30 years of Fragile X research, from identifying its cause, through finding dozens of treatment targets, through a series of disappointing clinical trials.
Read morePositive Results Reported in Phase II Fragile X Clinical Trial of PDE4D Inhibitor Zatolmilast from Tetra Therapeutics

Today, Tetra Therapeutics announces the first unequivocally positive phase 2 clinical trial in Fragile X syndrome, press release below. The results do not depend on carving out a subset of patients or post hoc analysis.
Read moreCompanies Move to Advance Potential Cognitive Treatment for Fragile X

Tetra Therapeutics and Shionogi announced plans to expand their partnership supporting BPN14770, a treatment candidate for disorders marked by cognitive and memory deficits, including Fragile X syndrome and Alzheimer’s disease. The agreement builds on an earlier collaboration between the two companies, and aims to further accelerate BPN14770’s development and potential marketing. It is currently in clinical testing in both Fragile X and Alzheimer’s patients.
Read moreShould You Participate in a Fragile X Clinical Trial?

A Fragile X clinical trial of a new PDE4D allosteric inhibitor from Tetra Therapeutics is nearly complete. Right now there are 3 remaining spots open to males 18-45 years of age with Fragile X syndrome. Dr. Elizabeth Berry-Kravis at the Rush University Medical Center in Chicago is leading this trial. The drug being studied has a unique mechanism of action that might improve cognitive and memory function.
Read moreScreening 2,320 FDA-Approved Drugs for Potential Treatment of Fragile X

FRAXA Research Foundation has awarded a $90,000 grant to Principal Investigator Dr. Sean McBride and Postdoctoral Fellow Dr. Karen Joyce, at Rowan University, to screen all 2,320 FDA-approved drugs on both mouse and fly models of Fragile X syndrome. Those drugs which show promise will be tested in more detail for potential to treat Fragile X in humans.
Read moreTetra Announces $40M to Advance BPN14770 for FXS and Alzheimer’s Disease

Tetra Discovery Partners has signed a multi-part deal that could bring it up to $160 million, plus royalties, from Shionogi & Co, Ltd, a Japanese major research-driven pharmaceutical company. Tetra currently is conducting an investigational Phase 2 study of BPN14770 in adults with Fragile X Syndrome, an indication for which BPN14770 has received Orphan Drug Designation from the US Food and Drug Administration. This clinical trial was made possible by early work with the FRAXA-DVI and over $200,000 from FRAXA.
Read moreResearch Points to Drugs which Inhibit PDE to Treat Fragile X

FRAXA Research Foundation funded a grant of $90,000 over 2016-2018, for a postdoctoral fellowship for Thomas Maurin, PhD, working under the mentorship of Dr. Barbara Bardoni at INSERM in France. The team works on the biochemistry of the Fragile X protein. They have found that PDE inhibitors (a class of drugs) show promise as treatments for Fragile X syndrome. In related research, FRAXA is currently funding a clinical trial of PDE4D inhibitors.
Read moreTetra Discovery Partners Initiates Phase 2 Trial of BPN14770 in Fragile X Syndrome

This 2-Period Crossover Study of BPN14770 is accepting adults males with Fragile X syndrome at Rush University Medical Center in Chicago. Principal Investigator of the study is Elizabeth Berry-Kravis, MD, PhD.
A selective inhibitor of the phosphodiesterase type-4D (PDE4D), BPN14770 has shown the ability to improve the quality of connections between neurons and to improve multiple behavioral outcomes in the Fragile X mouse model.
Combinatorial Drug Treatment in a Model of Fragile X Syndrome using Novel Biomarkers

With a $90,000 grant from FRAXA Research Foundation awarded over 2016-2017, University of California researchers Khaleel Razak, PhD, and Jonathan W. Lovelace, PhD, are exploring drug combinations to limit hypersensitivity to sounds in Fragile X mice.
Read moreEncouraging Results from First Trial of Minocycline in Fragile X

With a $40,000 grant from FRAXA, Dr. Carlo Paribello and his team at the Surrey Place Centre Fragile X clinic in Toronto, Ontario, ran an open label trial to see if minocycline can improve learning and reduce anxiety and behavioral problems in people with Fragile X. Twenty participants between the ages of 13 and 35 years took minocycline for two months.
Read moreRole of Matrix Metalloproteinases (MMP-9) in Fragile X

With a $220,000 grant from FRAXA Research Foundation over 3 years, Dr. Iryna Ethell from the University of California at Riverside studied the regulation of dendritic structure by matrix metalloproteinases and other extracellular signaling pathways. This work identified a major treatment strategy for Fragile X with the available MMP-9 inhibitor, minocycline.
Read more
