Opening the Gate: BK Channel Modulation as a Promising Treatment Strategy for Fragile X Syndrome
BK Channels: A Fragile X Treatment Target
A promising new treatment target has emerged from a series of FRAXA-funded research grants. These studies have built the case that BK channel function is impaired in Fragile X and that enhancing their activity with small-molecule openers is a potential approach to treating Fragile X syndrome.
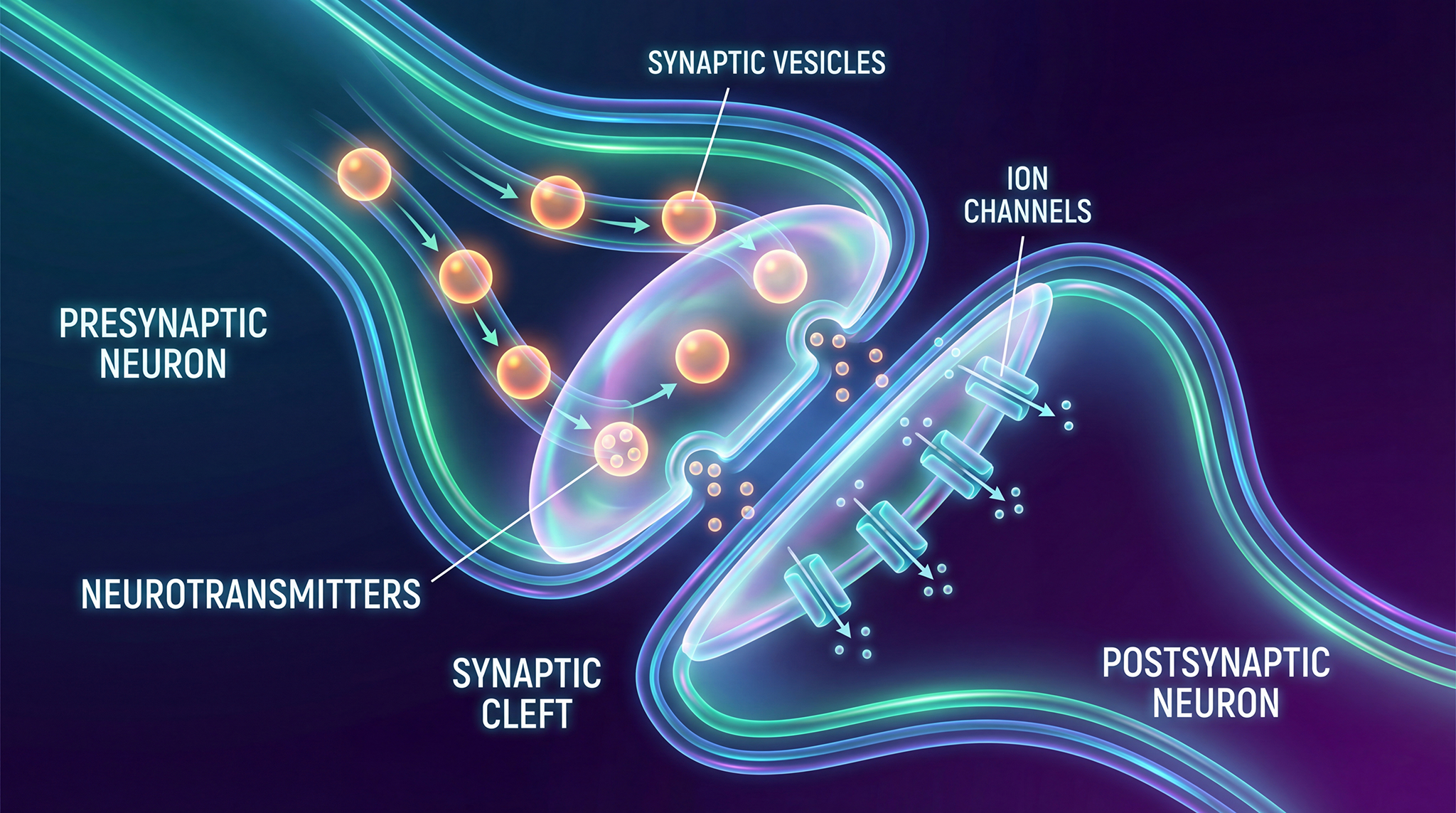
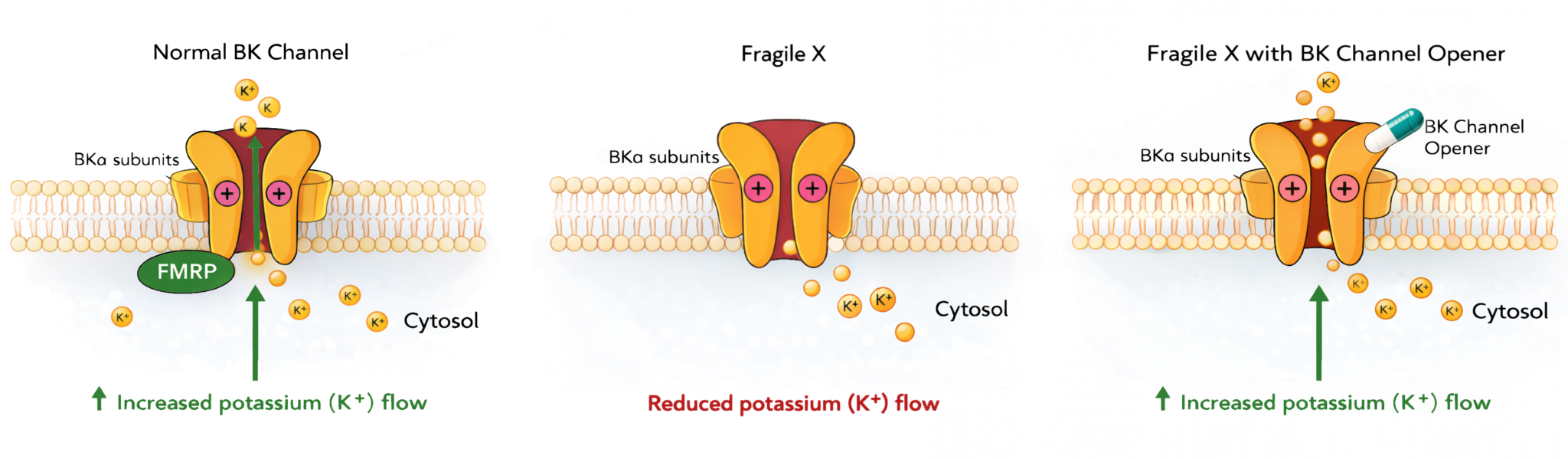
BK (Big Potassium) channels are potassium channels in neurons that act like adjustable gates and volume controls for brain activity. When calcium levels or electrical signals rise, they open to let potassium flow out, helping neurons quiet themselves and reset. This process disrupted in Fragile X syndrome.
What began as a biochemical observation in a lab has evolved into a promising therapeutic direction with a clear mechanistic chain, substantial preclinical validation, and clinical programs underway.
From Slack to BK: The Roots of Potassium Channel Research in Fragile X
The first serious suggestion that potassium channel dysfunction might play a central role in Fragile X pathophysiology came from the lab of Dr. Leonard Kaczmarek at Yale University. In a landmark study, Kaczmarek and colleagues discovered that FMRP binds directly to the C-terminal domain of the Slack potassium channel (also known as KNa1.1, a sodium-activated potassium channel), altering its gating properties.
In neurons, Slack channels play a role in action potential repolarization and the regulation of neuronal firing rates. FMRP's binding enhanced Slack channel activity, suggesting that one of FMRP’s functions is to modulate ion channel gating directly — not just indirectly through its well-known role in regulating mRNA translation.
This finding introduced a new therapeutic possibility: could potassium channel modulation compensate for FMRP loss in Fragile X syndrome?
Kaczmarek’s group and others began to explore that question, extending their investigations beyond Slack to include related potassium channels.
Proteomic Clues: BK Channels Are Reduced in Fragile X Brain
Support for this hypothesis emerged from an unexpected angle: proteomics. Dr. Peter Vanderklish and colleagues at Scripps applied advanced mass spectrometry techniques to study protein expression in the brains of Fmr1 knockout mice, the most widely used animal model of Fragile X syndrome.
Their data showed that levels of BK channel subunits were significantly reduced in key brain regions of Fragile X mice, including the hippocampus and cortex. Given BK channels' critical role in shaping action potentials and controlling synaptic excitability, this reduction suggested a functional consequence: loss of FMRP leads not only to dysregulated protein synthesis, but also to a deficit in a crucial mechanism for taming neuronal overactivity.
This finding reinforced the idea that BK channels could be a therapeutic target for Fragile X. These channels are regulated by FMRP and they are biochemically deficient in the absence of FMRP.
Why BK Channels? Advantages for Targeted Therapy
Following these discoveries, researchers began to focus increasingly on BK channels over Slack and other potassium channels. The rationale was compelling:
- BK channels are broadly expressed in the nervous system, especially in dendrites, axon initial segments, and presynaptic terminals, where they control firing precision and synaptic timing.
- They are activated by both voltage and intracellular calcium, allowing them to rapidly respond to neuronal activity and limit hyperexcitability.
- BK dysfunction has been implicated in epilepsy, auditory hypersensitivity, and gain control deficits—phenotypes that overlap significantly with those seen in Fragile X syndrome.
These properties made BK channels especially attractive as excitability stabilizers — an appealing target in a condition like Fragile X, where neuronal circuits are persistently overactive.
Genetic Evidence from Patients: BK Haploinsufficiency and Autism Spectrum Disorder (ASD)
Evidence for BK channel involvement in ASD has also come from the clinic. Dr. Sylvain Briault and colleagues at the University of Orléans identified a de novo loss-of-function mutation in KCNMA1, the gene encoding the BK channel’s alpha subunit, in a child with autism, intellectual disability, motor delays, and seizures.
This patient’s BK channel function was effectively halved — a condition known as haploinsufficiency. This rare but telling case demonstrated that disruption of BK channels is sufficient to produce an autism-like phenotype in humans, even in the absence of FMRP loss. It provided direct human genetic evidence that BK channel dysfunction could be causative — not merely secondary — in neurodevelopmental disorders.
Mechanistic Link: A Point Mutation in FMRP Disrupts BK Channel Binding
Additional clarity came from the Emory University lab of Dr. Stephen Warren, one of the original discoverers of the FMR1 gene. Warren’s team identified a missense mutation in a young male patient who exhibited Fragile X–like symptoms but did not carry the typical CGG repeat expansion that silences FMR1. Instead, the patient had a point mutation that disrupted the region of FMRP responsible for binding BK channels.
This unusual case suggested that FMRP’s ability to bind BK is critical for its normal function, and that selective disruption of this interaction is sufficient to produce a Fragile X–like disorder, despite otherwise intact FMRP expression.
As a remarkable validation and extension of this hypothesis, Dr. Vitaly Klyachko’s team at Washington University performed detailed electrophysiological experiments. Dr. Klyachko had been investigating BK channels for many years and had already identified abnormailities in Fragile X. They demonstrated that wild-type FMRP enhances BK channel activation, while mutant FMRP (bearing the patient’s point mutation) does not. Without this regulation, neurons become hyperexcitable, firing more frequently and less precisely, which contributes to impaired sensory processing and cognition.
This research closed the mechanistic loop: FMRP binds and modulates BK channels. When this interaction is lost — whether due to FMRP loss, FMRP mutation, or BK haploinsufficiency — neuronal excitability spirals out of control.
Preclinical Proof: BK Channel Openers Rescue Fragile X Phenotypes in Mice
NOTE: look for graphs/images from their paper.
Armed with this mechanistic foundation, researchers turned to the obvious next question: what happens if you pharmacologically boost BK channel activity in Fragile X models?
In a series of studies led by Dr. Jacques Pichon and Dr. Sylvain Briault, researchers at CNRS and the University of Orléans tested BK channel openers in Fmr1 knockout mice.
Their results were striking: BK activators reversed key behavioral abnormalities in Fragile X mice, including:
- Hyperactivity
- Anxiety-like behaviors
- Impaired cognitive performance
- Aberrant sensory processing, particularly in visual discrimination tasks
In one study, Fragile X mice treated with a BK opener showed improved object recognition and sensory responses, suggesting that BK modulation can restore both behavioral and perceptual function.
Other groups have since replicated these findings using additional BK channel activators, including chlorzoxazone, a well-known muscle relaxant with BK-activating properties.
Into the Clinic: BK Channel Modulators in Human Trials
(With strong preclinical results in hand, clinical development is now underway:
- Servier acquired Kaerus Bioscience’s BK channel opener (in a deal potentially worth $450 million), which has shown safety and brain engagement in Phase 1 studies and is moving into Phase 2.
- Spinogenix is advancing SPG601, a BK channel opener, in Phase 2 trials for Fragile X syndrome.
- Repurposing efforts are also exploring whether existing drugs with BK-opening properties, like chlorzoxazone, could offer benefit in FXS or other neurodevelopmental disorders.
Challenges remain: BK channels are expressed throughout the body, so achieving CNS-specific modulation is essential to avoid side effects. Identifying biomarkers of target engagement (e.g., EEG or fMRI signatures) will also be key in guiding dose and monitoring response.
Conclusion: Converging Lines of Evidence
Few therapeutic strategies in Fragile X are supported by such a tight, multi-tiered chain of evidence. The story of BK channel modulation brings together:
- Molecular biology: FMRP directly binds and regulates BK channels.
- Proteomics: BK channels are underexpressed in Fmr1-deficient brains.
- Human genetics: Mutations in both FMR1 and KCNMA1 cause overlapping ASD phenotypes.
- Electrophysiology: FMRP loss disrupts BK-dependent firing precision.
- Behavioral neuroscience: BK openers rescue Fragile X phenotypes in mice.
- Clinical development: Drug candidates are now in clinical trials to treat Fragile X syndrome.
If these efforts succeed, BK channel openers may become the first targeted treatment for Fragile X syndrome that addresses its core excitability dysfunction at the presynaptic level.
What started as an obscure interaction between a protein and a potassium channel could open new doors for families living with Fragile X — and perhaps illuminate broader therapeutic paths for other autism spectrum disorders driven by neuronal hyperexcitability.
Written by
Michael Tranfaglia, MD
Medical Director, Treasurer, Co-Founder
Dr. Michael Tranfaglia is Medical Director and Chief Scientific Officer of FRAXA Research Foundation, coordinating the Foundation’s research strategy and working with university and industry scientists to develop new therapeutic agents for Fragile X. He has a BA in Biology from Harvard University and an MD from the University of North Carolina at Chapel Hill. His son Andy has Fragile X syndrome.