FRAXADev – Developing BK Channel Openers for Fragile X Syndrome

A number of people have asked us about FRAXADev, a new project starting in France; this is a nonprofit initiative which seeks to develop a new kind of drug for Fragile X. The drugs they are interested in testing in Fragile X clinical trials were developed by Bristol-Myers Squibb many years ago, and are now off patent. This class of drugs opens a potassium channel in the membrane of neurons, which helps to decrease neuronal excitability. These agents are called “BK Channel Openers”. The project is seeking donations to this effort, but they are not a 501(c)3 tax-exempt organization, so for US residents this is mostly a “crowdfunding” appeal at this point.
There are a few points we’d like to clarify, since so many people are asking:
- FRAXADev isn’t part of FRAXA Research Foundation; these are two completely different organizations.
- The research team was originally funded by FRAXA, but the name is coincidental.
- We have been asked to provide advice to this project, and have been in contact with the research group; however, we have no financial relationship with FRAXADev.
- It’s good science, and BK channel openers are a reasonable thing to try for Fragile X.
- This research team has obtained some promising preclinical results which appear to justify clinical trials.
- We don’t think it is reasonable or realistic to try to fund this kind of drug development project with donations from the Fragile X community or general public—this is the appropriate role of pharmaceutical companies, which have much greater access to capital.
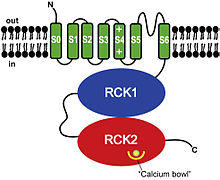
About BK Channel Openers
Written by
Michael Tranfaglia, MD
Medical Director, Treasurer, Co-Founder
Dr. Michael Tranfaglia is Medical Director and Chief Scientific Officer of FRAXA Research Foundation, coordinating the Foundation’s research strategy and working with university and industry scientists to develop new therapeutic agents for Fragile X. He has a BA in Biology from Harvard University and an MD from the University of North Carolina at Chapel Hill. His son Andy has Fragile X syndrome.

